Injectable Biomaterial & Hydrogel Systems
Injectable Biomaterial and Hydrogel Systems
Injectable Biomaterial and Hydrogel Systems
We are highly active in developing injectable biomaterial systems for different biomedical applications. They are highly attractive for musculoskeletal tissue repair as they:
1.Provide cell protection during needle delivery;
2.Prevent cell leakage;
3.Provides a protective/instructive niche (cell anchorage/differentiation);
4.Facilitate advanced strategies (e.g. growth factors/agents/gene delivery)
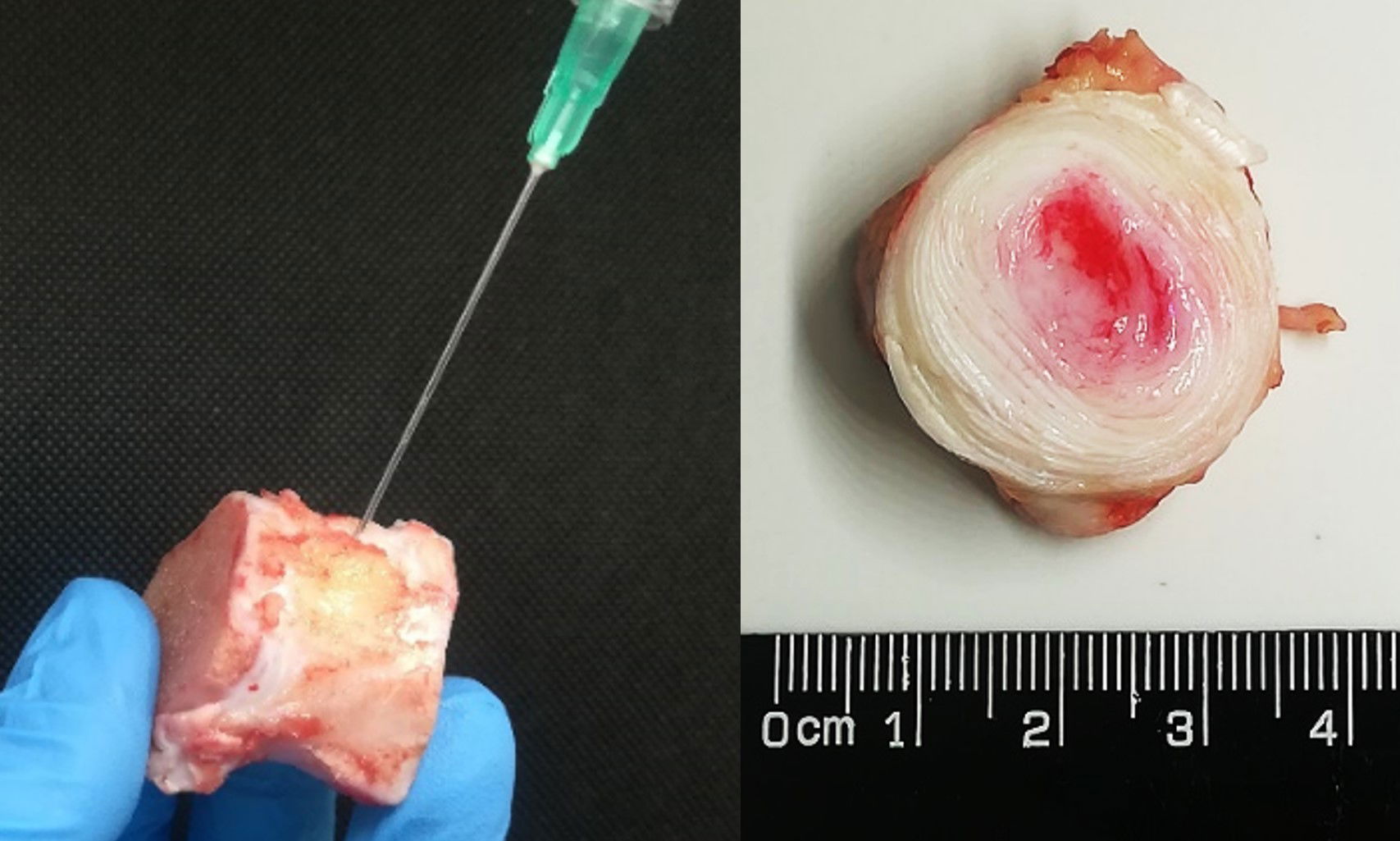
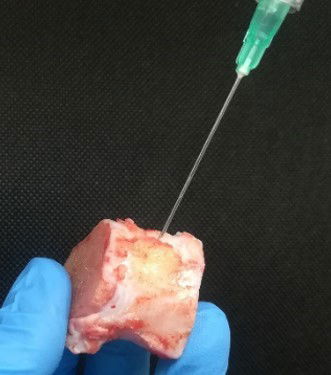
Figure: Injectable disc derived extracellular matrix hydrogels chemically functionalised with chondroitin sulfate
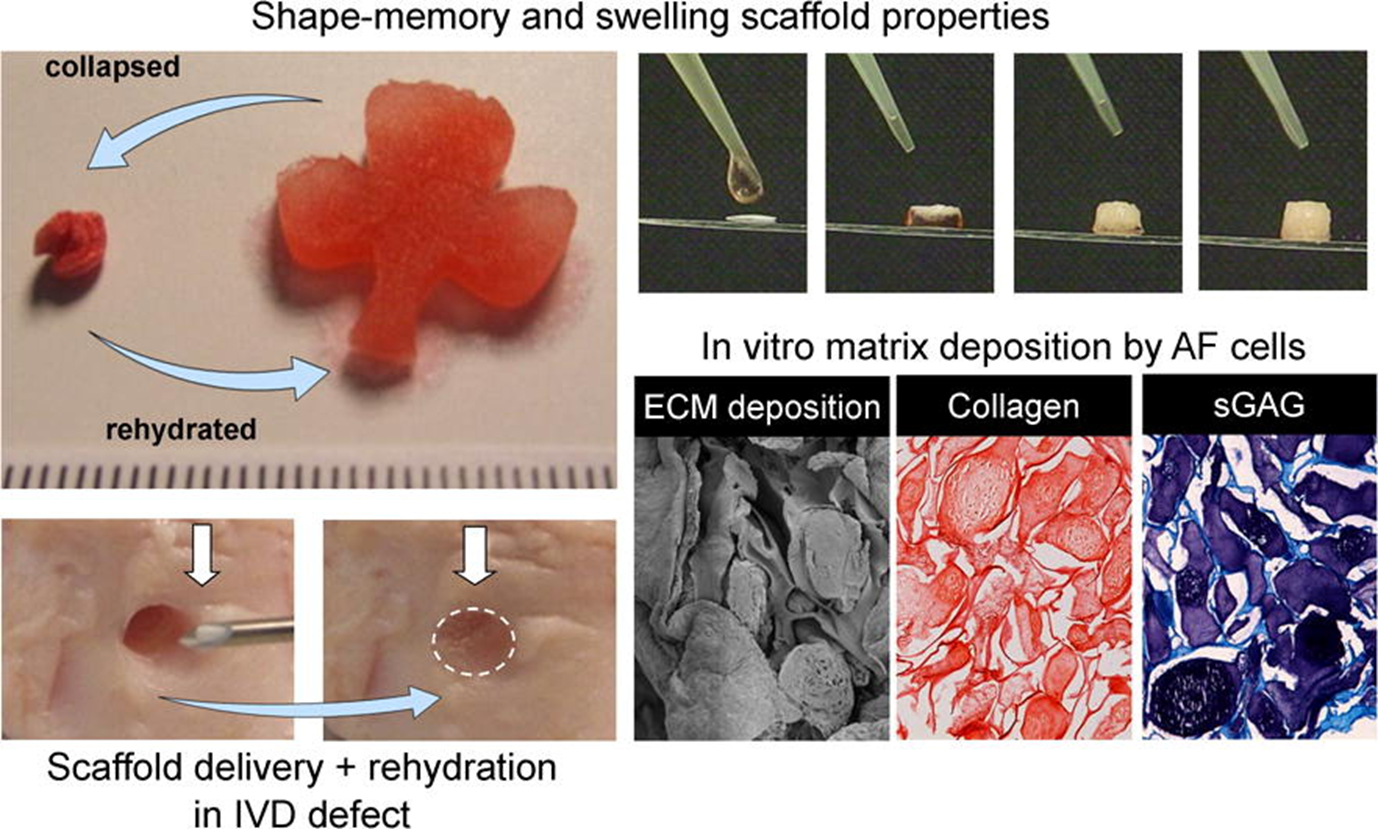
Figure: Injectable shape memory porous alginate scaffolds can be delivered minimally invasively and promote matrix formation.
Guillaume, O., Daly, A., Lennon K., Gansau J., Buckley S.F. and Buckley, C.T. Shape-memory porous alginate scaffolds for regeneration of the annulus fibrosus - Effect of TGF-β3 supplementation and oxygen culture conditions. Acta Biomaterialia 10: 1985–1995, 2014.
Self Healing Hydrogel Systems
These are a branch of hydrogel systems which can undergo “self-healing” or repair. Networks through dynamic Schiff-base- continuous breaking and regenerating resulting in injectable and self-healing properties

Injectable microcapsules and microcarriers
Recent work in our lab has focused on developing injectable microcapsules and microcarriers that can be primed in vitro using growth factors
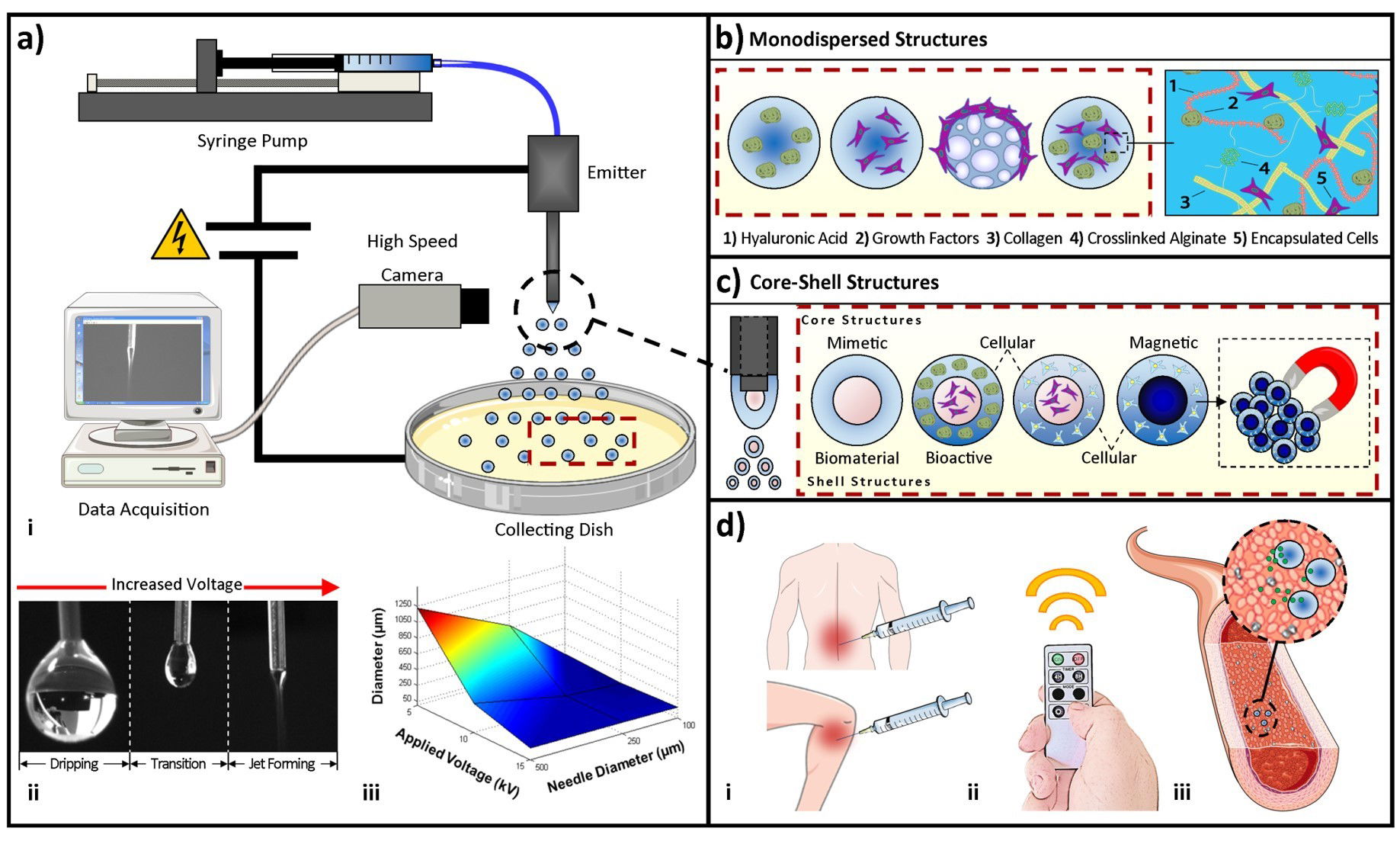
Naqvi S.M., Vedicherla S.V., Gansau J, McIntyre T, Doherty M and Buckley C.T. “Living Cell Factories”- Electrosprayed Microcapsules and Microcarriers for Minimally Invasive Delivery. Advanced Materials, 28(27):5662-71, 2016.
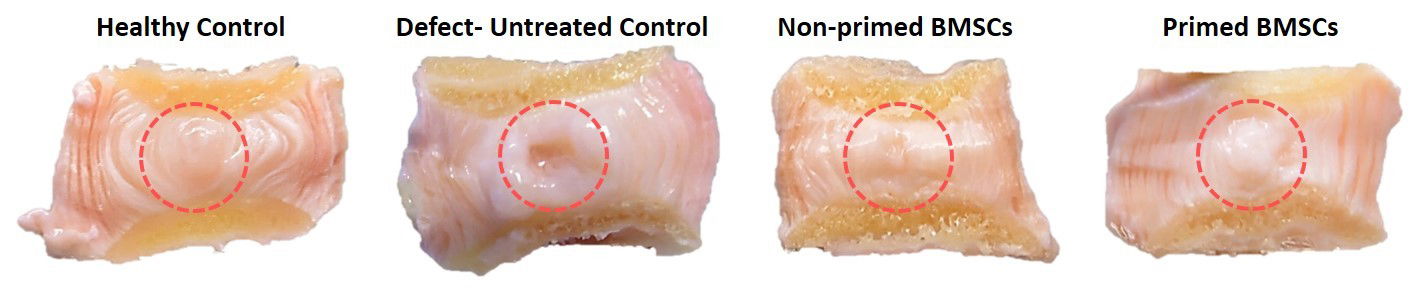
Macroscopic images of healthy, untreated, unprimed and primed microencapsulated BMSC-treated bovine discs after 28 d of whole organ culture demonstrating tissue regeneration
Naqvi S.M., Gansau J., Gibbons, D.L. and Buckley C.T. Primed and cryopreserved stem cell microcapsules for IVD regeneration - in vitro co-culture and ex vivo organ culture assessment. eCM 37: 134-152, 2019


